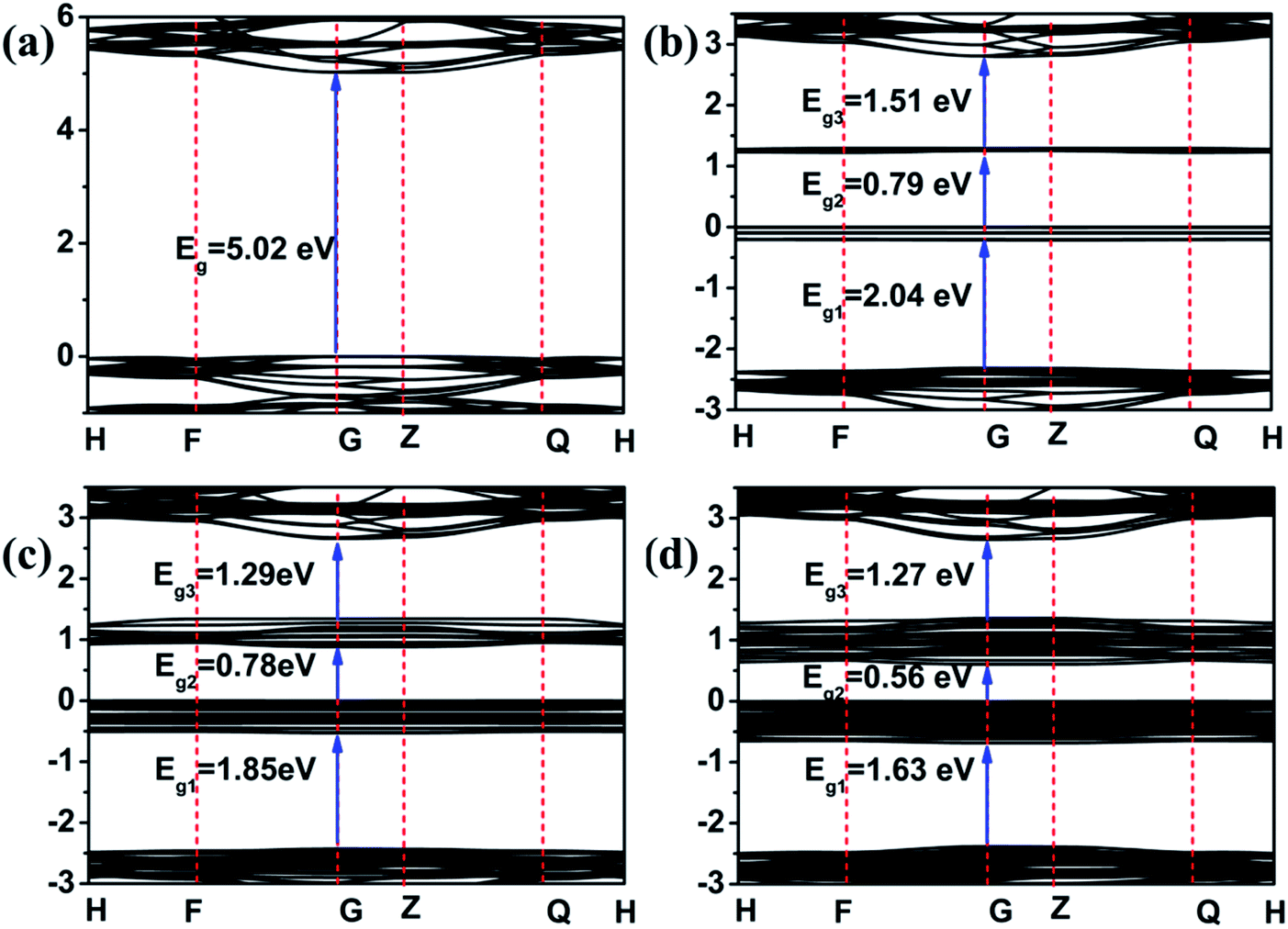
Photocatalytic degradation of diphenhydramine in aqueous solution by natural dolomite - RSC Advances (RSC Publishing) DOI:10.1039/D0RA07533G

On the interaction between calcite and dolomite: Insights from gas and aqueous geochemistry and mineralogical characterization - ScienceDirect

Self-accelerating dolomite-for-calcite replacement: Self-organized dynamics of burial dolomitization and associated mineralization | American Journal of Science
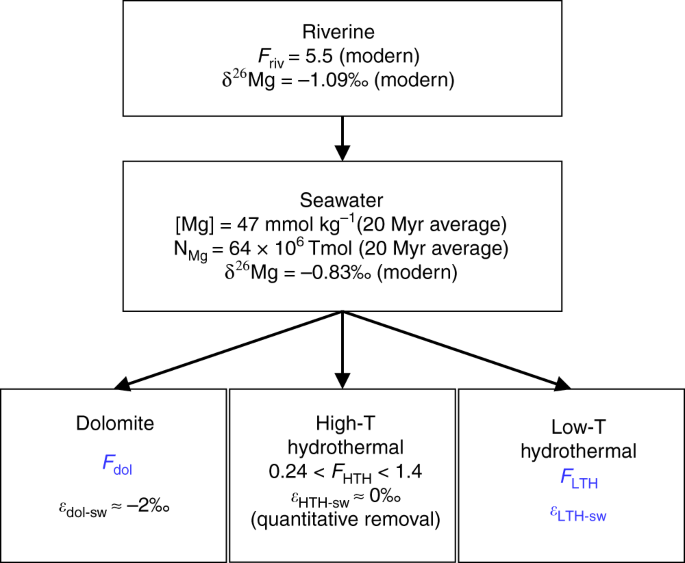
New isotope constraints on the Mg oceanic budget point to cryptic modern dolomite formation | Nature Communications

Bed‐scale spatial patterns in dolomite abundance: Part II. Effect of varied fluid chemistry, flow rate, precursor mineralogy, temperature, textural heterogeneity, nucleation density and bed geometry - Budd - 2019 - Sedimentology - Wiley Online Library

Bed‐scale spatial patterns in dolomite abundance: Part II. Effect of varied fluid chemistry, flow rate, precursor mineralogy, temperature, textural heterogeneity, nucleation density and bed geometry - Budd - 2019 - Sedimentology - Wiley Online Library
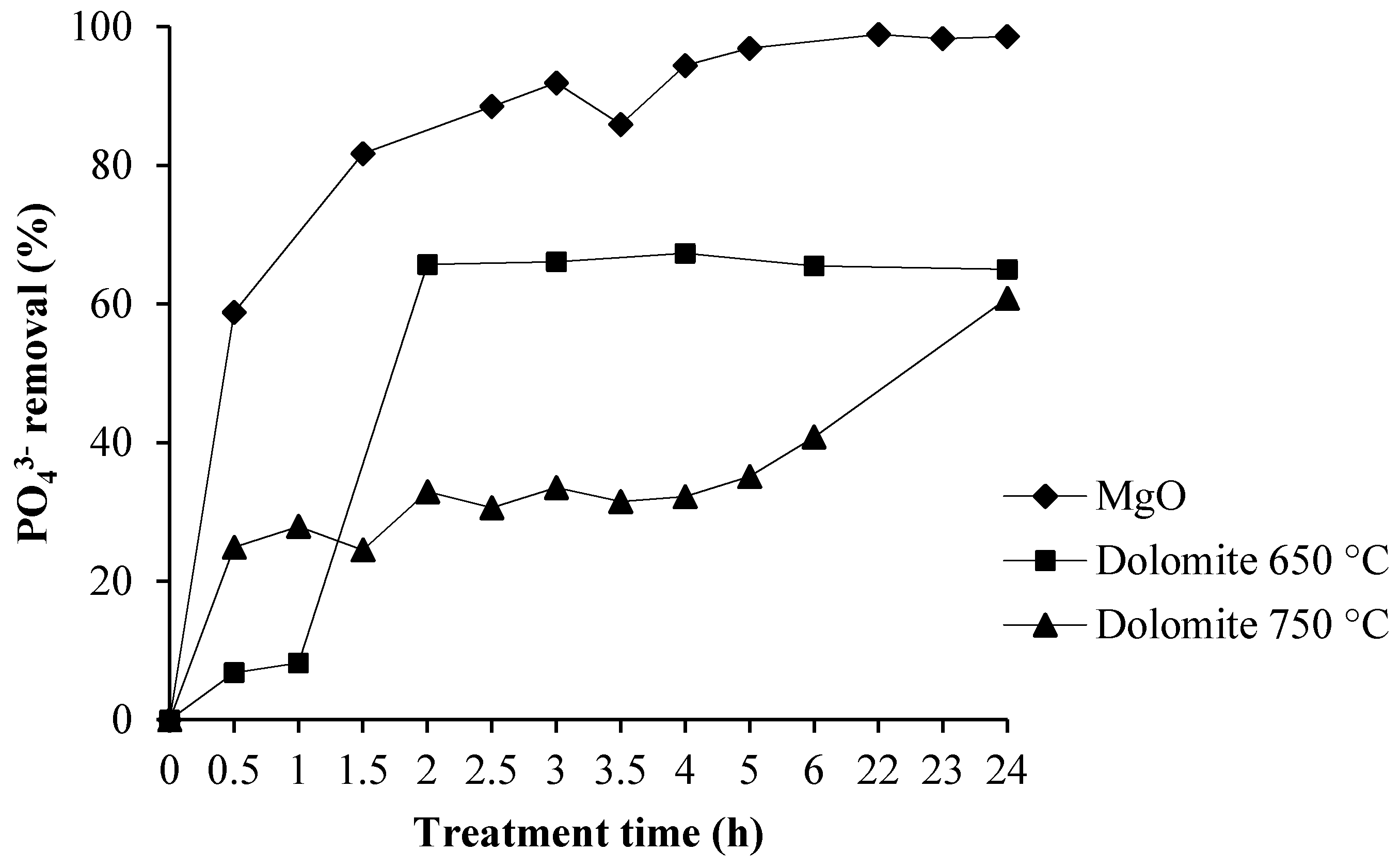
ChemEngineering | Free Full-Text | Use of Calcined Dolomite as Chemical Precipitant in the Simultaneous Removal of Ammonium and Phosphate from Synthetic Wastewater and from Agricultural Sludge | HTML
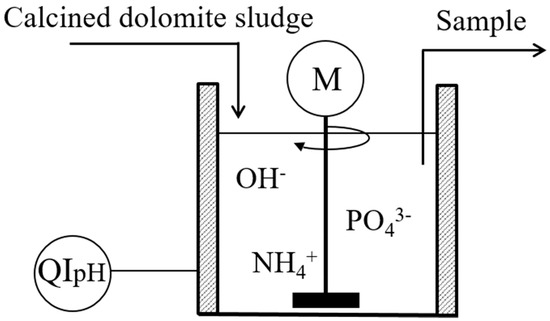
ChemEngineering | Free Full-Text | Use of Calcined Dolomite as Chemical Precipitant in the Simultaneous Removal of Ammonium and Phosphate from Synthetic Wastewater and from Agricultural Sludge | HTML

Time-resolved high-temperature Raman spectra of dolomite at 9 m in the... | Download Scientific Diagram
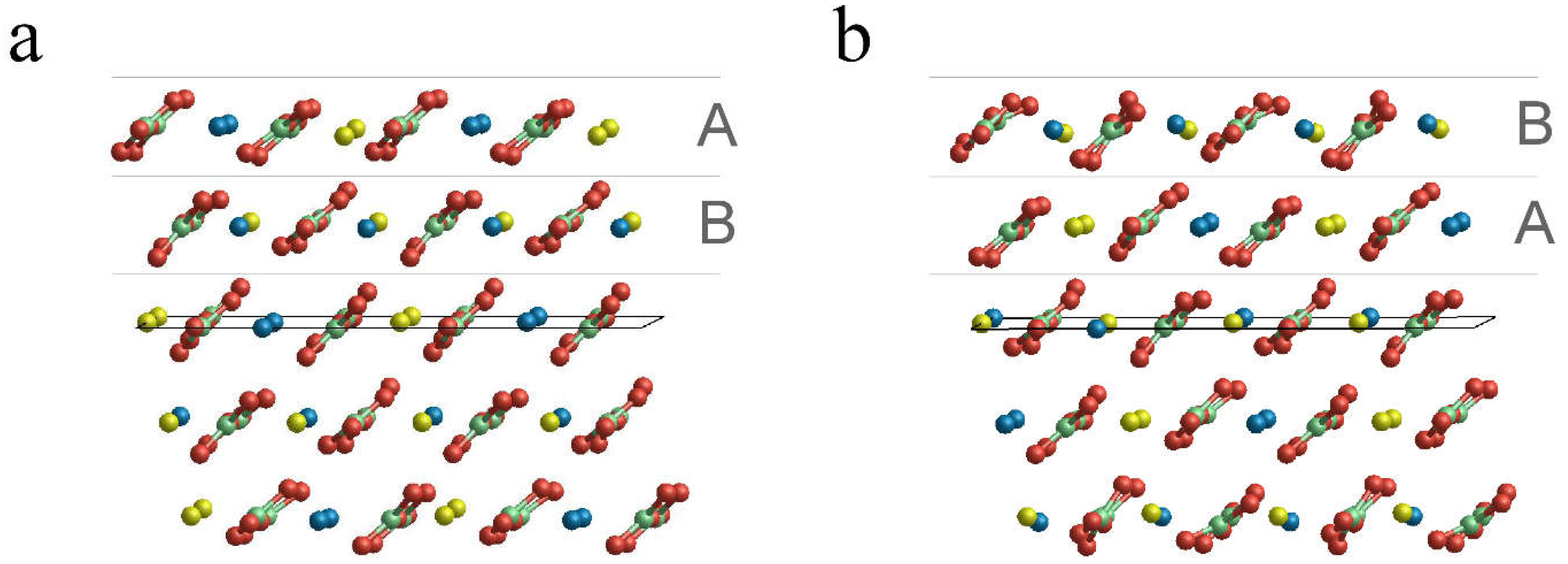
Minerals | Free Full-Text | (10.4) Face of Ordered and Disordered Dolomite, MgCa(CO3)2: A Computational Study to Reveal the Growth Mechanism | HTML












